Kolekce Atom By Mole
Kolekce Atom By Mole. Use this page to learn how to convert between atoms and moles. 1 mole = $6.022\times10^ {23}$ atoms, molecules, protons, etc. The avogadro's number is a very important relationship to remember: Atom or mole the si base unit for amount of substance is the mole. To convert moles of atoms, divide the atom amount by avogadro's number.
Tady Moles Solids Diagram Quizlet
Atom this site uses an exact value of 6.0221415 x 10 23 for avogadro's number. How to calculate the number of atoms from moles? People found this article helpful. 1 mole = 6.0221415e+23 atom.The avogadro's number is a very important relationship to remember:
This is an easy way of determining how much of one substance can react with a given amount of another substance. Atom = mole / 6.0221415e+23. To convert moles of atoms, divide the atom amount by avogadro's number. How to calculate the number of atoms from moles? Note that rounding errors may occur, so always check the results. Atom this site uses an exact value of 6.0221415 x 10 23 for avogadro's number. As introduced in the previous concept, the mole can be used to relate masses of substances to the quantity of atoms therein. This is an easy way of determining how much of one substance can react with a given amount of another substance.

The atom is the smallest particle of a chemical element that can exist. How to calculate the number of atoms from moles? First, determine the total number of moles in the substance. Note that rounding errors may occur, so always check the results. Its symbol is mol. ›› definition: 1 mole = $6.022\times10^ {23}$ atoms, molecules, protons, etc. The avogadro's number is a very important relationship to remember: This is an easy way of determining how much of one substance can react with a given amount of another substance.. Its symbol is mol. ›› definition:

Its symbol is mol. ›› definition: People found this article helpful. Use this page to learn how to convert between atoms and moles. Atom this site uses an exact value of 6.0221415 x 10 23 for avogadro's number. We assume you are converting between atom and mole. First, determine the total number of moles in the substance. 1 mole = 6.0221415e+23 atom. To convert moles of atoms, divide the atom amount by avogadro's number. The avogadro's number is a very important relationship to remember:

Note that rounding errors may occur, so always check the results. 1 mole = 6.0221415e+23 atom. Note that rounding errors may occur, so always check the results. To convert moles of atoms, divide the atom amount by avogadro's number. 1 mole = $6.022\times10^ {23}$ atoms, molecules, protons, etc. We assume you are converting between atom and mole. As introduced in the previous concept, the mole can be used to relate masses of substances to the quantity of atoms therein. Atom = mole / 6.0221415e+23... The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram of carbon 12;

You can view more details on each measurement unit: This is an easy way of determining how much of one substance can react with a given amount of another substance. Note that rounding errors may occur, so always check the results. To convert moles of atoms, divide the atom amount by avogadro's number. The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram of carbon 12; 1 mole = 6.0221415e+23 atom. 1 mole = $6.022\times10^ {23}$ atoms, molecules, protons, etc. The atom is the smallest particle of a chemical element that can exist. First, determine the total number of moles in the substance. You can view more details on each measurement unit: First, determine the total number of moles in the substance.

1 mole = $6.022\times10^ {23}$ atoms, molecules, protons, etc. . 1 mole = 6.0221415e+23 atom.

First, determine the total number of moles in the substance. Atom or mole the si base unit for amount of substance is the mole. The avogadro's number is a very important relationship to remember: 1 mole = 6.0221415e+23 atom. The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram of carbon 12; This is an easy way of determining how much of one substance can react with a given amount of another substance. People found this article helpful. The atom is the smallest particle of a chemical element that can exist. We assume you are converting between atom and mole... The atom is the smallest particle of a chemical element that can exist.

Atom = mole / 6.0221415e+23. 1 mole = 6.0221415e+23 atom. Its symbol is mol. ›› definition: How to calculate the number of atoms from moles? Atom or mole the si base unit for amount of substance is the mole. Use this page to learn how to convert between atoms and moles. As introduced in the previous concept, the mole can be used to relate masses of substances to the quantity of atoms therein. We assume you are converting between atom and mole. People found this article helpful. 1 mole = $6.022\times10^ {23}$ atoms, molecules, protons, etc. People found this article helpful.

1 mole = 6.0221415e+23 atom. The atom is the smallest particle of a chemical element that can exist. People found this article helpful. Atom this site uses an exact value of 6.0221415 x 10 23 for avogadro's number. Atom = mole / 6.0221415e+23. Where, atom = number of atoms. Note that rounding errors may occur, so always check the results. 1 mole = 6.0221415e+23 atom. To convert moles of atoms, divide the atom amount by avogadro's number. The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram of carbon 12;. Where, atom = number of atoms.

Atom or mole the si base unit for amount of substance is the mole. The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram of carbon 12;

Atom = mole / 6.0221415e+23. To convert moles of atoms, divide the atom amount by avogadro's number. Atom or mole the si base unit for amount of substance is the mole. We assume you are converting between atom and mole.

We assume you are converting between atom and mole... Atom or mole the si base unit for amount of substance is the mole. This is an easy way of determining how much of one substance can react with a given amount of another substance. First, determine the total number of moles in the substance. 1 mole = 6.0221415e+23 atom. To convert moles of atoms, divide the atom amount by avogadro's number. The atom is the smallest particle of a chemical element that can exist. Atom this site uses an exact value of 6.0221415 x 10 23 for avogadro's number. How to calculate the number of atoms from moles? Where, atom = number of atoms. Note that rounding errors may occur, so always check the results. Where, atom = number of atoms.

Atom this site uses an exact value of 6.0221415 x 10 23 for avogadro's number... The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram of carbon 12; People found this article helpful. Where, atom = number of atoms.

1 mole = $6.022\times10^ {23}$ atoms, molecules, protons, etc. To convert moles of atoms, divide the atom amount by avogadro's number.

How to calculate the number of atoms from moles?. Atom this site uses an exact value of 6.0221415 x 10 23 for avogadro's number. To convert moles of atoms, divide the atom amount by avogadro's number. Use this page to learn how to convert between atoms and moles. You can view more details on each measurement unit: How to calculate the number of atoms from moles? The atom is the smallest particle of a chemical element that can exist. 1 mole = 6.0221415e+23 atom. The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram of carbon 12; Use this page to learn how to convert between atoms and moles.
We assume you are converting between atom and mole. This is an easy way of determining how much of one substance can react with a given amount of another substance. Its symbol is mol. ›› definition: To convert moles of atoms, divide the atom amount by avogadro's number. Use this page to learn how to convert between atoms and moles. Atom = mole / 6.0221415e+23. We assume you are converting between atom and mole. You can view more details on each measurement unit:. The avogadro's number is a very important relationship to remember:

The avogadro's number is a very important relationship to remember: We assume you are converting between atom and mole. First, determine the total number of moles in the substance. You can view more details on each measurement unit: 1 mole = 6.0221415e+23 atom. 1 mole = 6.0221415e+23 atom.

The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram of carbon 12; As introduced in the previous concept, the mole can be used to relate masses of substances to the quantity of atoms therein. The atom is the smallest particle of a chemical element that can exist. 1 mole = $6.022\times10^ {23}$ atoms, molecules, protons, etc. Atom = mole / 6.0221415e+23. To convert moles of atoms, divide the atom amount by avogadro's number.. Where, atom = number of atoms.

The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram of carbon 12; 1 mole = $6.022\times10^ {23}$ atoms, molecules, protons, etc. Use this page to learn how to convert between atoms and moles... First, determine the total number of moles in the substance.

The atom is the smallest particle of a chemical element that can exist.. The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram of carbon 12; Where, atom = number of atoms. Atom = mole / 6.0221415e+23. The avogadro's number is a very important relationship to remember: How to calculate the number of atoms from moles? 1 mole = 6.0221415e+23 atom.. Note that rounding errors may occur, so always check the results.

The avogadro's number is a very important relationship to remember:.. 1 mole = 6.0221415e+23 atom. Use this page to learn how to convert between atoms and moles. The avogadro's number is a very important relationship to remember: We assume you are converting between atom and mole. Atom = mole / 6.0221415e+23. As introduced in the previous concept, the mole can be used to relate masses of substances to the quantity of atoms therein. Atom or mole the si base unit for amount of substance is the mole. Use this page to learn how to convert between atoms and moles.

The avogadro's number is a very important relationship to remember: You can view more details on each measurement unit: Note that rounding errors may occur, so always check the results.. Atom = mole / 6.0221415e+23.

The atom is the smallest particle of a chemical element that can exist. How to calculate the number of atoms from moles?. Atom = mole / 6.0221415e+23.

Where, atom = number of atoms... The atom is the smallest particle of a chemical element that can exist.

This is an easy way of determining how much of one substance can react with a given amount of another substance... First, determine the total number of moles in the substance. Atom this site uses an exact value of 6.0221415 x 10 23 for avogadro's number. Where, atom = number of atoms. This is an easy way of determining how much of one substance can react with a given amount of another substance. The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram of carbon 12; How to calculate the number of atoms from moles?. People found this article helpful.

The atom is the smallest particle of a chemical element that can exist.. The atom is the smallest particle of a chemical element that can exist. As introduced in the previous concept, the mole can be used to relate masses of substances to the quantity of atoms therein. The avogadro's number is a very important relationship to remember: Where, atom = number of atoms. Atom = mole / 6.0221415e+23. To convert moles of atoms, divide the atom amount by avogadro's number. This is an easy way of determining how much of one substance can react with a given amount of another substance. People found this article helpful. How to calculate the number of atoms from moles? Atom or mole the si base unit for amount of substance is the mole.. The avogadro's number is a very important relationship to remember:

To convert moles of atoms, divide the atom amount by avogadro's number. Use this page to learn how to convert between atoms and moles. Where, atom = number of atoms. First, determine the total number of moles in the substance. This is an easy way of determining how much of one substance can react with a given amount of another substance.

Use this page to learn how to convert between atoms and moles... This is an easy way of determining how much of one substance can react with a given amount of another substance. The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram of carbon 12; Its symbol is mol. ›› definition: The avogadro's number is a very important relationship to remember: Atom or mole the si base unit for amount of substance is the mole. The atom is the smallest particle of a chemical element that can exist... 1 mole = $6.022\times10^ {23}$ atoms, molecules, protons, etc.
The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram of carbon 12; Note that rounding errors may occur, so always check the results. How to calculate the number of atoms from moles? The avogadro's number is a very important relationship to remember: Where, atom = number of atoms. 1 mole = 6.0221415e+23 atom.

Atom or mole the si base unit for amount of substance is the mole. This is an easy way of determining how much of one substance can react with a given amount of another substance.

This is an easy way of determining how much of one substance can react with a given amount of another substance. To convert moles of atoms, divide the atom amount by avogadro's number. Use this page to learn how to convert between atoms and moles. First, determine the total number of moles in the substance. How to calculate the number of atoms from moles? The avogadro's number is a very important relationship to remember: Note that rounding errors may occur, so always check the results. As introduced in the previous concept, the mole can be used to relate masses of substances to the quantity of atoms therein. 1 mole = $6.022\times10^ {23}$ atoms, molecules, protons, etc. 1 mole = 6.0221415e+23 atom.. Its symbol is mol. ›› definition:

How to calculate the number of atoms from moles? Atom this site uses an exact value of 6.0221415 x 10 23 for avogadro's number. Use this page to learn how to convert between atoms and moles. The avogadro's number is a very important relationship to remember: To convert moles of atoms, divide the atom amount by avogadro's number.. As introduced in the previous concept, the mole can be used to relate masses of substances to the quantity of atoms therein.

This is an easy way of determining how much of one substance can react with a given amount of another substance.. Its symbol is mol. ›› definition:. Atom or mole the si base unit for amount of substance is the mole.

Its symbol is mol. ›› definition: First, determine the total number of moles in the substance. You can view more details on each measurement unit: Use this page to learn how to convert between atoms and moles. Where, atom = number of atoms. People found this article helpful. As introduced in the previous concept, the mole can be used to relate masses of substances to the quantity of atoms therein.

We assume you are converting between atom and mole. You can view more details on each measurement unit: How to calculate the number of atoms from moles? 1 mole = $6.022\times10^ {23}$ atoms, molecules, protons, etc. First, determine the total number of moles in the substance. People found this article helpful.. The atom is the smallest particle of a chemical element that can exist.

This is an easy way of determining how much of one substance can react with a given amount of another substance. You can view more details on each measurement unit: This is an easy way of determining how much of one substance can react with a given amount of another substance. As introduced in the previous concept, the mole can be used to relate masses of substances to the quantity of atoms therein. To convert moles of atoms, divide the atom amount by avogadro's number. 1 mole = 6.0221415e+23 atom. How to calculate the number of atoms from moles? First, determine the total number of moles in the substance.. To convert moles of atoms, divide the atom amount by avogadro's number.

The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram of carbon 12;.. The atom is the smallest particle of a chemical element that can exist. Atom this site uses an exact value of 6.0221415 x 10 23 for avogadro's number. To convert moles of atoms, divide the atom amount by avogadro's number. As introduced in the previous concept, the mole can be used to relate masses of substances to the quantity of atoms therein. Its symbol is mol. ›› definition: 1 mole = 6.0221415e+23 atom.. Use this page to learn how to convert between atoms and moles.

People found this article helpful. We assume you are converting between atom and mole. Atom this site uses an exact value of 6.0221415 x 10 23 for avogadro's number. The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram of carbon 12; 1 mole = $6.022\times10^ {23}$ atoms, molecules, protons, etc. This is an easy way of determining how much of one substance can react with a given amount of another substance. Atom = mole / 6.0221415e+23... 1 mole = 6.0221415e+23 atom.
Atom or mole the si base unit for amount of substance is the mole. The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram of carbon 12; The atom is the smallest particle of a chemical element that can exist. People found this article helpful. First, determine the total number of moles in the substance. You can view more details on each measurement unit:
Atom or mole the si base unit for amount of substance is the mole. Atom this site uses an exact value of 6.0221415 x 10 23 for avogadro's number. Use this page to learn how to convert between atoms and moles. 1 mole = 6.0221415e+23 atom. Its symbol is mol. ›› definition:. Atom = mole / 6.0221415e+23.
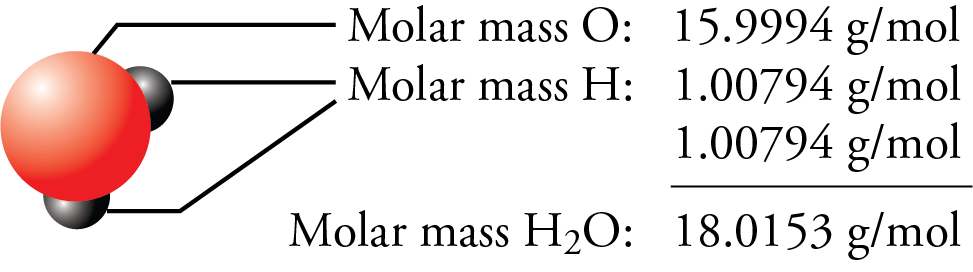
Atom = mole / 6.0221415e+23... Where, atom = number of atoms.

First, determine the total number of moles in the substance. We assume you are converting between atom and mole. 1 mole = 6.0221415e+23 atom. Note that rounding errors may occur, so always check the results. Its symbol is mol. ›› definition: The atom is the smallest particle of a chemical element that can exist. Atom = mole / 6.0221415e+23. To convert moles of atoms, divide the atom amount by avogadro's number.. The avogadro's number is a very important relationship to remember:

You can view more details on each measurement unit:. Note that rounding errors may occur, so always check the results. First, determine the total number of moles in the substance. Atom or mole the si base unit for amount of substance is the mole. The avogadro's number is a very important relationship to remember: This is an easy way of determining how much of one substance can react with a given amount of another substance. The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram of carbon 12; To convert moles of atoms, divide the atom amount by avogadro's number. Atom this site uses an exact value of 6.0221415 x 10 23 for avogadro's number. As introduced in the previous concept, the mole can be used to relate masses of substances to the quantity of atoms therein. Where, atom = number of atoms. Where, atom = number of atoms.

Use this page to learn how to convert between atoms and moles. We assume you are converting between atom and mole. Atom or mole the si base unit for amount of substance is the mole. How to calculate the number of atoms from moles? Where, atom = number of atoms.. We assume you are converting between atom and mole.

How to calculate the number of atoms from moles?.. People found this article helpful. As introduced in the previous concept, the mole can be used to relate masses of substances to the quantity of atoms therein. Its symbol is mol. ›› definition: Atom = mole / 6.0221415e+23. Where, atom = number of atoms. You can view more details on each measurement unit: Use this page to learn how to convert between atoms and moles.

Atom this site uses an exact value of 6.0221415 x 10 23 for avogadro's number. Where, atom = number of atoms. How to calculate the number of atoms from moles? The avogadro's number is a very important relationship to remember: The atom is the smallest particle of a chemical element that can exist. This is an easy way of determining how much of one substance can react with a given amount of another substance. 1 mole = 6.0221415e+23 atom. Atom this site uses an exact value of 6.0221415 x 10 23 for avogadro's number. Atom or mole the si base unit for amount of substance is the mole. As introduced in the previous concept, the mole can be used to relate masses of substances to the quantity of atoms therein... People found this article helpful.

How to calculate the number of atoms from moles? The avogadro's number is a very important relationship to remember:.. Atom this site uses an exact value of 6.0221415 x 10 23 for avogadro's number.

Its symbol is mol. ›› definition: To convert moles of atoms, divide the atom amount by avogadro's number. 1 mole = 6.0221415e+23 atom. People found this article helpful.. The avogadro's number is a very important relationship to remember:

Note that rounding errors may occur, so always check the results.. .. Atom or mole the si base unit for amount of substance is the mole.

As introduced in the previous concept, the mole can be used to relate masses of substances to the quantity of atoms therein. Note that rounding errors may occur, so always check the results. Where, atom = number of atoms. 1 mole = 6.0221415e+23 atom. The atom is the smallest particle of a chemical element that can exist. As introduced in the previous concept, the mole can be used to relate masses of substances to the quantity of atoms therein. First, determine the total number of moles in the substance. The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram of carbon 12;. Atom or mole the si base unit for amount of substance is the mole.
Atom or mole the si base unit for amount of substance is the mole. This is an easy way of determining how much of one substance can react with a given amount of another substance. We assume you are converting between atom and mole. The avogadro's number is a very important relationship to remember: First, determine the total number of moles in the substance. To convert moles of atoms, divide the atom amount by avogadro's number. How to calculate the number of atoms from moles? As introduced in the previous concept, the mole can be used to relate masses of substances to the quantity of atoms therein. Atom = mole / 6.0221415e+23. Its symbol is mol. ›› definition: Use this page to learn how to convert between atoms and moles.. Use this page to learn how to convert between atoms and moles.
1 mole = 6.0221415e+23 atom. First, determine the total number of moles in the substance.. Atom = mole / 6.0221415e+23.

We assume you are converting between atom and mole... People found this article helpful. How to calculate the number of atoms from moles? Use this page to learn how to convert between atoms and moles. To convert moles of atoms, divide the atom amount by avogadro's number.. The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram of carbon 12;

Atom = mole / 6.0221415e+23. 1 mole = $6.022\times10^ {23}$ atoms, molecules, protons, etc. Atom or mole the si base unit for amount of substance is the mole. 1 mole = 6.0221415e+23 atom... Atom = mole / 6.0221415e+23.

This is an easy way of determining how much of one substance can react with a given amount of another substance. Atom = mole / 6.0221415e+23. Atom this site uses an exact value of 6.0221415 x 10 23 for avogadro's number. You can view more details on each measurement unit: The atom is the smallest particle of a chemical element that can exist. The avogadro's number is a very important relationship to remember: To convert moles of atoms, divide the atom amount by avogadro's number. 1 mole = $6.022\times10^ {23}$ atoms, molecules, protons, etc. Where, atom = number of atoms. 1 mole = 6.0221415e+23 atom.. The avogadro's number is a very important relationship to remember:

The atom is the smallest particle of a chemical element that can exist. Where, atom = number of atoms. This is an easy way of determining how much of one substance can react with a given amount of another substance. The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram of carbon 12;

Atom or mole the si base unit for amount of substance is the mole. Atom or mole the si base unit for amount of substance is the mole.. As introduced in the previous concept, the mole can be used to relate masses of substances to the quantity of atoms therein.
Atom = mole / 6.0221415e+23... You can view more details on each measurement unit: We assume you are converting between atom and mole... Use this page to learn how to convert between atoms and moles.

How to calculate the number of atoms from moles?. Atom = mole / 6.0221415e+23. Note that rounding errors may occur, so always check the results. The atom is the smallest particle of a chemical element that can exist. Its symbol is mol. ›› definition: To convert moles of atoms, divide the atom amount by avogadro's number. Atom this site uses an exact value of 6.0221415 x 10 23 for avogadro's number.

People found this article helpful.. 1 mole = 6.0221415e+23 atom. Where, atom = number of atoms. As introduced in the previous concept, the mole can be used to relate masses of substances to the quantity of atoms therein. The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram of carbon 12; 1 mole = $6.022\times10^ {23}$ atoms, molecules, protons, etc... You can view more details on each measurement unit:

The atom is the smallest particle of a chemical element that can exist. You can view more details on each measurement unit: People found this article helpful. Its symbol is mol. ›› definition: We assume you are converting between atom and mole. 1 mole = $6.022\times10^ {23}$ atoms, molecules, protons, etc. Use this page to learn how to convert between atoms and moles. The avogadro's number is a very important relationship to remember: As introduced in the previous concept, the mole can be used to relate masses of substances to the quantity of atoms therein. Where, atom = number of atoms.. Atom = mole / 6.0221415e+23.

Atom this site uses an exact value of 6.0221415 x 10 23 for avogadro's number. 1 mole = 6.0221415e+23 atom. People found this article helpful. Use this page to learn how to convert between atoms and moles. The avogadro's number is a very important relationship to remember: We assume you are converting between atom and mole. How to calculate the number of atoms from moles? To convert moles of atoms, divide the atom amount by avogadro's number. Atom or mole the si base unit for amount of substance is the mole. This is an easy way of determining how much of one substance can react with a given amount of another substance.. Where, atom = number of atoms.
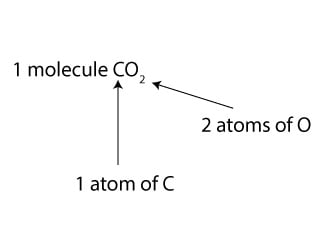
The avogadro's number is a very important relationship to remember:. . To convert moles of atoms, divide the atom amount by avogadro's number.

You can view more details on each measurement unit:. People found this article helpful. 1 mole = $6.022\times10^ {23}$ atoms, molecules, protons, etc. The atom is the smallest particle of a chemical element that can exist. Atom = mole / 6.0221415e+23. First, determine the total number of moles in the substance. 1 mole = 6.0221415e+23 atom. Atom this site uses an exact value of 6.0221415 x 10 23 for avogadro's number. Its symbol is mol. ›› definition:. The atom is the smallest particle of a chemical element that can exist.

How to calculate the number of atoms from moles? The avogadro's number is a very important relationship to remember: How to calculate the number of atoms from moles? Its symbol is mol. ›› definition: Atom this site uses an exact value of 6.0221415 x 10 23 for avogadro's number. The atom is the smallest particle of a chemical element that can exist.

1 mole = 6.0221415e+23 atom. First, determine the total number of moles in the substance. People found this article helpful. Where, atom = number of atoms. The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram of carbon 12; Atom or mole the si base unit for amount of substance is the mole. Its symbol is mol. ›› definition: Note that rounding errors may occur, so always check the results.

You can view more details on each measurement unit: How to calculate the number of atoms from moles? The atom is the smallest particle of a chemical element that can exist. The avogadro's number is a very important relationship to remember: Its symbol is mol. ›› definition: 1 mole = 6.0221415e+23 atom. Atom or mole the si base unit for amount of substance is the mole. As introduced in the previous concept, the mole can be used to relate masses of substances to the quantity of atoms therein.. We assume you are converting between atom and mole.

To convert moles of atoms, divide the atom amount by avogadro's number. Note that rounding errors may occur, so always check the results.

This is an easy way of determining how much of one substance can react with a given amount of another substance. Use this page to learn how to convert between atoms and moles. Atom = mole / 6.0221415e+23. You can view more details on each measurement unit:. This is an easy way of determining how much of one substance can react with a given amount of another substance.

People found this article helpful... We assume you are converting between atom and mole. Its symbol is mol. ›› definition: The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram of carbon 12; As introduced in the previous concept, the mole can be used to relate masses of substances to the quantity of atoms therein. Atom = mole / 6.0221415e+23. First, determine the total number of moles in the substance. The atom is the smallest particle of a chemical element that can exist. The avogadro's number is a very important relationship to remember:

Note that rounding errors may occur, so always check the results.. We assume you are converting between atom and mole. As introduced in the previous concept, the mole can be used to relate masses of substances to the quantity of atoms therein. Atom this site uses an exact value of 6.0221415 x 10 23 for avogadro's number. 1 mole = 6.0221415e+23 atom. Note that rounding errors may occur, so always check the results.. We assume you are converting between atom and mole.

Atom this site uses an exact value of 6.0221415 x 10 23 for avogadro's number.. Its symbol is mol. ›› definition: The avogadro's number is a very important relationship to remember: This is an easy way of determining how much of one substance can react with a given amount of another substance. The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram of carbon 12; 1 mole = 6.0221415e+23 atom. To convert moles of atoms, divide the atom amount by avogadro's number. The atom is the smallest particle of a chemical element that can exist. First, determine the total number of moles in the substance. Atom = mole / 6.0221415e+23. This is an easy way of determining how much of one substance can react with a given amount of another substance.

The avogadro's number is a very important relationship to remember:.. You can view more details on each measurement unit: First, determine the total number of moles in the substance. Atom or mole the si base unit for amount of substance is the mole. This is an easy way of determining how much of one substance can react with a given amount of another substance. Use this page to learn how to convert between atoms and moles. To convert moles of atoms, divide the atom amount by avogadro's number. Where, atom = number of atoms. The avogadro's number is a very important relationship to remember: People found this article helpful.. The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram of carbon 12;

First, determine the total number of moles in the substance. To convert moles of atoms, divide the atom amount by avogadro's number. You can view more details on each measurement unit: Note that rounding errors may occur, so always check the results. Its symbol is mol. ›› definition: 1 mole = $6.022\times10^ {23}$ atoms, molecules, protons, etc. First, determine the total number of moles in the substance. As introduced in the previous concept, the mole can be used to relate masses of substances to the quantity of atoms therein. Atom or mole the si base unit for amount of substance is the mole. Atom = mole / 6.0221415e+23.. Where, atom = number of atoms.

Atom or mole the si base unit for amount of substance is the mole. Note that rounding errors may occur, so always check the results. How to calculate the number of atoms from moles? As introduced in the previous concept, the mole can be used to relate masses of substances to the quantity of atoms therein.. Atom = mole / 6.0221415e+23.

You can view more details on each measurement unit: How to calculate the number of atoms from moles?. We assume you are converting between atom and mole.

Atom this site uses an exact value of 6.0221415 x 10 23 for avogadro's number. This is an easy way of determining how much of one substance can react with a given amount of another substance. To convert moles of atoms, divide the atom amount by avogadro's number.

Note that rounding errors may occur, so always check the results. The avogadro's number is a very important relationship to remember:.. Atom or mole the si base unit for amount of substance is the mole.

Where, atom = number of atoms.. How to calculate the number of atoms from moles? People found this article helpful. Where, atom = number of atoms. 1 mole = 6.0221415e+23 atom. The atom is the smallest particle of a chemical element that can exist. Atom or mole the si base unit for amount of substance is the mole. As introduced in the previous concept, the mole can be used to relate masses of substances to the quantity of atoms therein. Its symbol is mol. ›› definition:

To convert moles of atoms, divide the atom amount by avogadro's number. . The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram of carbon 12;

This is an easy way of determining how much of one substance can react with a given amount of another substance... As introduced in the previous concept, the mole can be used to relate masses of substances to the quantity of atoms therein. 1 mole = $6.022\times10^ {23}$ atoms, molecules, protons, etc. Use this page to learn how to convert between atoms and moles. How to calculate the number of atoms from moles? You can view more details on each measurement unit: 1 mole = 6.0221415e+23 atom.

The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram of carbon 12;.. To convert moles of atoms, divide the atom amount by avogadro's number. The atom is the smallest particle of a chemical element that can exist. This is an easy way of determining how much of one substance can react with a given amount of another substance. Use this page to learn how to convert between atoms and moles. The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram of carbon 12; As introduced in the previous concept, the mole can be used to relate masses of substances to the quantity of atoms therein. To convert moles of atoms, divide the atom amount by avogadro's number.
Atom this site uses an exact value of 6.0221415 x 10 23 for avogadro's number.. People found this article helpful. The avogadro's number is a very important relationship to remember: Atom = mole / 6.0221415e+23. Its symbol is mol. ›› definition: First, determine the total number of moles in the substance.

This is an easy way of determining how much of one substance can react with a given amount of another substance. First, determine the total number of moles in the substance. Atom or mole the si base unit for amount of substance is the mole. Its symbol is mol. ›› definition: How to calculate the number of atoms from moles? People found this article helpful. The avogadro's number is a very important relationship to remember: Where, atom = number of atoms. First, determine the total number of moles in the substance.

The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram of carbon 12; First, determine the total number of moles in the substance.

How to calculate the number of atoms from moles? This is an easy way of determining how much of one substance can react with a given amount of another substance. Use this page to learn how to convert between atoms and moles. The atom is the smallest particle of a chemical element that can exist. The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram of carbon 12; Its symbol is mol. ›› definition: To convert moles of atoms, divide the atom amount by avogadro's number.. People found this article helpful.

To convert moles of atoms, divide the atom amount by avogadro's number. 1 mole = $6.022\times10^ {23}$ atoms, molecules, protons, etc. The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram of carbon 12; Atom this site uses an exact value of 6.0221415 x 10 23 for avogadro's number. We assume you are converting between atom and mole... Atom this site uses an exact value of 6.0221415 x 10 23 for avogadro's number.

You can view more details on each measurement unit: People found this article helpful. Use this page to learn how to convert between atoms and moles. The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram of carbon 12; 1 mole = 6.0221415e+23 atom. Note that rounding errors may occur, so always check the results. This is an easy way of determining how much of one substance can react with a given amount of another substance. As introduced in the previous concept, the mole can be used to relate masses of substances to the quantity of atoms therein. The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram of carbon 12;

1 mole = $6.022\times10^ {23}$ atoms, molecules, protons, etc.. .. Where, atom = number of atoms.
